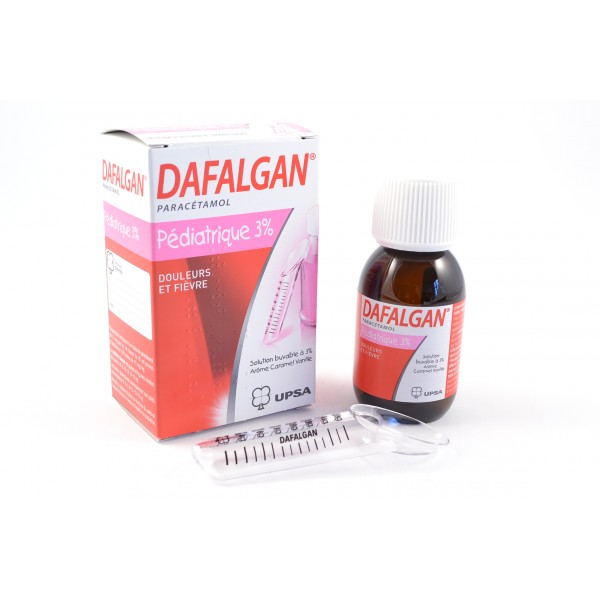
Luxembourg's Ministry of Health has announced that Pediatric Dafalgan (30 mg/ml) has been withdrawn from sale across the Grand Duchy; it is used to treat Fever, Muscle Pain, Pain and Sciatica.
The firm, Bristol-Myers Squibb Belgium, has withdrawn the medicine due to non-compliance as evidence was discovered of polyurethane particles in the packaging equipment. The investigation has not ruled out that only one batch has been contaminated.
Only certain lot numbers are affected; anyone who may have purchased Pediatric Dafalgan 30 mg/ml CNK 1571 314 (90 ml bottle oral solution), with any of the following lot numbers, is asked to return them to the pharmacy from which they obtained them: N9631, P0311, P1063, P1228, P2000, P2506, P2799, P4104, P4949, P6300, P5862, P7008, P8288, P9471, R0035, R1214, R2165, R2692, R3181, R4302, R4304.
Titulaire d’enregistrement: Bristol-Myers Squibb Belgium








